Keywords
KRAS
аллостерическое ингибирование
дизайн лекарств
молекулярное моделирование
панселективность
рак легкого
Abstract
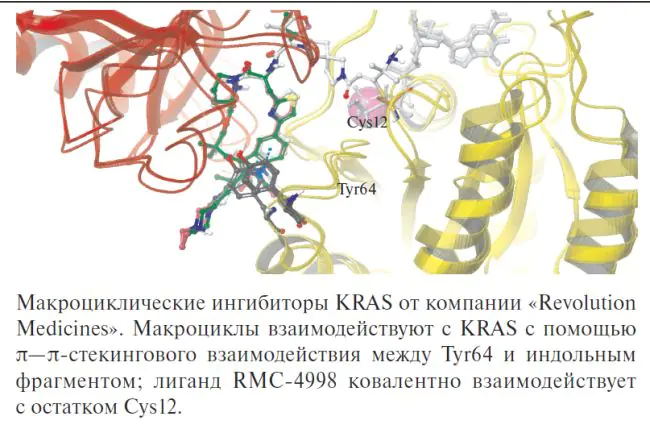
Представлен подробный обзор последних достижений в разработке ингибиторов мутантных форм гуанозинтрифосфатазы KRAS, которая играет ключевую роль в патогенезе различных онкологических заболеваний. Сделан акцент на сложности и ограничениях таргетирования KRAS из-за его структурных особенностей и высокой частоты мутаций. Основное внимание уделено панселективным ингибиторам, способным действовать на несколько различных мутаций KRAS, что важно для преодоления резистентности опухолей к терапии. Приведены примеры перспективных молекулярных структур, анализ механизмов действия и возможных причин селективности.
References
1.

Bos J.L., Rehmann H., Wittinghofer A.
Cell,
2007
2.

Vigil D., Cherfils J., Rossman K.L., Der C.J.
Nature Reviews Cancer,
2010
3.

Malumbres M., Barbacid M.
Nature Reviews Cancer,
2003
4.

Downward J.
Nature Reviews Cancer,
2003
5.

Moore A.R., Rosenberg S.C., McCormick F., Malek S.
Nature Reviews Drug Discovery,
2020
6.

Kim D., Xue J.Y., Lito P.
Cell,
2020
7.

Huang L., Guo Z., Wang F., Fu L.
Signal Transduction and Targeted Therapy,
2021
8.

Wittinghofer A., Vetter I.R.
Annual Review of Biochemistry,
2011
9.

Abraham S.J., Muhamed I., Nolet R., Yeung F., Gaponenko V.
Protein Expression and Purification,
2010
10.

Czyzyk D., Yan W., Messing S., Gillette W., Tsuji T., Yamaguchi M., Furuzono S., Turner D.M., Esposito D., Nissley D.V., McCormick F., Simanshu D.K.
Nature Communications,
2025
11.

Kwan A.K., Piazza G.A., Keeton A.B., Leite C.A.
Journal of Experimental and Clinical Cancer Research,
2022
12.

Dang C.V., Reddy E.P., Shokat K.M., Soucek L.
Nature Reviews Cancer,
2017
13.
Bagchi S., Rathee P., Jayaprakash V., Banerjee S.
Mini-Reviews in Medicinal Chemistry,
2018
14.

Nussinov R., Jang H., Tsai C., Liao T., Li S., Fushman D., Zhang J.
Cellular and Molecular Life Sciences,
2017
15.

Hampton S.E., Dore T.M., Schmidt W.K.
Critical Reviews in Biochemistry and Molecular Biology,
2018
16.

Diver M.M., Pedi L., Koide A., Koide S., Long S.B.
Nature,
2018
17.

Mazieres J., Pradines A., Favre G.
Cancer Letters,
2004
18.

Whyte D.B., Kirschmeier P., Hockenberry T.N., Nunez-Oliva I., James L., Catino J.J., Bishop W.R., Pai J.
Journal of Biological Chemistry,
1997
19.

Court H., Amoyel M., Hackman M., Lee K.E., Xu R., Miller G., Bar-Sagi D., Bach E.A., Bergö M.O., Philips M.R.
Journal of Clinical Investigation,
2013
20.

Wahlstrom A.M., Cutts B.A., Karlsson C., Andersson K.M., Liu M., Sjogren A.M., Swolin B., Young S.G., Bergo M.O.
Blood,
2006
21.

Ou S.I., Koczywas M., Ulahannan S., Janne P., Pacheco J., Burris H., McCoach C., Wang J.S., Gordon M., Haura E., Riess J.W., Zhu V., Ng K., Eckhardt S.G., Capasso A., et. al.
Journal of Thoracic Oncology,
2020
22.

Nichols R.J., Haderk F., Stahlhut C., Schulze C.J., Hemmati G., Wildes D., Tzitzilonis C., Mordec K., Marquez A., Romero J., Hsieh T., Zaman A., Olivas V., McCoach C., Blakely C.M., et. al.
Nature Cell Biology,
2018
23.

Hofmann M.H., Gmachl M., Ramharter J., Savarese F., Gerlach D., Marszalek J.R., Sanderson M.P., Kessler D., Trapani F., Arnhof H., Rumpel K., Botesteanu D., Ettmayer P., Gerstberger T., Kofink C., et. al.
Cancer Discovery,
2021
24.

Zhu C., Guan X., Zhang X., Luan X., Song Z., Cheng X., Zhang W., Qin J.
Molecular Cancer,
2022
25.

Lito P., Rosen N., Solit D.B.
Nature Medicine,
2013
26.

Desai J., Gan H., Barrow C., Jameson M., Atkinson V., Haydon A., Millward M., Begbie S., Brown M., Markman B., Patterson W., Hill A., Horvath L., Nagrial A., Richardson G., et. al.
Journal of Clinical Oncology,
2020
27.

Kidger A.M., Sipthorp J., Cook S.J.
Pharmacology and Therapeutics,
2018
28.

Niemantsverdriet M., Schuuring E., Elst A.T., van der Wekken A.J., van Kempen L.C., van den Berg A., Groen H.J.
Journal of Thoracic Oncology,
2018
29.

Adachi Y., Kimura R., Hirade K., Ebi H.
Cancers,
2021
30.

Cook S.J., Lochhead P.A.
Frontiers in Cell and Developmental Biology,
2022
31.

Prior I.A., Hood F.E., Hartley J.L.
Cancer Research,
2020
32.

André F., Arnedos M., Baras A.S., Baselga J., Bedard P.L., Berger M.F., Bierkens M., Calvo F., Cerami E., Chakravarty D., Dang K.K., Davidson N.E., Del Vecchio Fitz C., Dogan S., DuBois R.N., et. al.
Cancer Discovery,
2017
33.

Ostrem J.M., Peters U., Sos M.L., Wells J.A., Shokat K.M.
Nature,
2013
34.

Kettle J.G., Cassar D.J.
Expert Opinion on Therapeutic Patents,
2020
35.

Xu Q., Zhang G., Liu Q., Li S., Zhang Y.
Expert Opinion on Therapeutic Patents,
2022
36.

Fell J.B., Fischer J.P., Baer B.R., Blake J.F., Bouhana K., Briere D.M., Brown K.D., Burgess L.E., Burns A.C., Burkard M.R., Chiang H., Chicarelli M.J., Cook A.W., Gaudino J.J., Hallin J., et. al.
Journal of Medicinal Chemistry,
2020
37.

Lanman B.A., Allen J.R., Allen J.G., Amegadzie A.K., Ashton K.S., Booker S.K., Chen J.J., Chen N., Frohn M.J., Goodman G., Kopecky D.J., Liu L., Lopez P., Low J.D., Ma V., et. al.
Journal of Medicinal Chemistry,
2019
38.

39.

40.

Nagasaka M., Li Y., Sukari A., Ou S.I., Al-Hallak M.N., Azmi A.S.
Cancer Treatment Reviews,
2020
41.

42.

Zheng Q., Zhang Z., Guiley K.Z., Shokat K.M.
Nature Chemical Biology,
2024
43.

Zhang Z., Morstein J., Ecker A.K., Guiley K.Z., Shokat K.M.
Journal of the American Chemical Society,
2022
44.

Zhang Z., Guiley K.Z., Shokat K.M.
Nature Chemical Biology,
2022
45.

Simultaneous Covalent Modification of K-Ras(G12D) and K-Ras(G12C) with Tunable Oxirane Electrophiles
Yu Z., He X., Wang R., Xu X., Zhang Z., Ding K., Zhang Z., Tan Y., Li Z.
Journal of the American Chemical Society,
2023
46.

Di Federico A., Ricciotti I., Favorito V., Michelina S.V., Scaparone P., Metro G., De Giglio A., Pecci F., Lamberti G., Ambrogio C., Ricciuti B.
Current Oncology Reports,
2023
47.

Awad M.M., Liu S., Rybkin I.I., Arbour K.C., Dilly J., Zhu V.W., Johnson M.L., Heist R.S., Patil T., Riely G.J., Jacobson J.O., Yang X., Persky N.S., Root D.E., Lowder K.E., et. al.
New England Journal of Medicine,
2021
48.

Liu J., Kang R., Tang D.
Cancer Gene Therapy,
2021
49.

Wang X., Allen S., Blake J.F., Bowcut V., Briere D.M., Calinisan A., Dahlke J.R., Fell J.B., Fischer J.P., Gunn R.J., Hallin J., Laguer J., Lawson J.D., Medwid J., Newhouse B., et. al.
Journal of Medicinal Chemistry,
2021
50.

Vasta J.D., Peacock D.M., Zheng Q., Walker J.A., Zhang Z., Zimprich C.A., Thomas M.R., Beck M.T., Binkowski B.F., Corona C.R., Robers M.B., Shokat K.M.
Nature Chemical Biology,
2022
51.

Hallin J., Bowcut V., Calinisan A., Briere D.M., Hargis L., Engstrom L.D., Laguer J., Medwid J., Vanderpool D., Lifset E., Trinh D., Hoffman N., Wang X., David Lawson J., Gunn R.J., et. al.
Nature Medicine,
2022
52.

Wang X.
Journal of Chemical Information and Modeling,
2021
53.

Keats M., Han J.J., Lee Y., Lee C., Luo J.
Cancer Research,
2023
54.

Mao Z., Xiao H., Shen P., Yang Y., Xue J., Yang Y., Shang Y., Zhang L., Li X., Zhang Y., Du Y., Chen C., Guo R., Zhang Y.
Cell Discovery,
2022
55.

Pantsar T.
Computational and Structural Biotechnology Journal,
2020
56.

Kim D., Herdeis L., Rudolph D., Zhao Y., Böttcher J., Vides A., Ayala-Santos C.I., Pourfarjam Y., Cuevas-Navarro A., Xue J.Y., Mantoulidis A., Bröker J., Wunberg T., Schaaf O., Popow J., et. al.
Nature,
2023
57.

Welsch M.E., Kaplan A., Chambers J.M., Stokes M.E., Bos P.H., Zask A., Zhang Y., Sanchez-Martin M., Badgley M.A., Huang C.S., Tran T.H., Akkiraju H., Brown L.M., Nandakumar R., Cremers S., et. al.
Cell,
2017
58.

McCarthy M.J., Pagba C.V., Prakash P., Naji A.K., van der Hoeven D., Liang H., Gupta A.K., Zhou Y., Cho K., Hancock J.F., Gorfe A.A.
ACS Omega,
2019
59.

Kessler D., Gmachl M., Mantoulidis A., Martin L.J., Zoephel A., Mayer M., Gollner A., Covini D., Fischer S., Gerstberger T., Gmaschitz T., Goodwin C., Greb P., Häring D., Hela W., et. al.
Proceedings of the National Academy of Sciences of the United States of America,
2019
60.

Kessler D., Gollner A., Gmachl M., Mantoulidis A., Martin L.J., Zoephel A., Mayer M., Covini D., Fischer S., Gerstberger T., Gmaschitz T., Goodwin C., Greb P., Häring D., Hela W., et. al.
Proceedings of the National Academy of Sciences of the United States of America,
2020
61.

Feng H., Zhang Y., Bos P.H., Chambers J.M., Dupont M.M., Stockwell B.R.
Biochemistry,
2019
62.

Tanaka T., Thomas J., Van Montfort R., Miller A., Rabbitts T.
Scientific Reports,
2021
63.

Quevedo C.E., Cruz-Migoni A., Bery N., Miller A., Tanaka T., Petch D., Bataille C.J., Lee L.Y., Fallon P.S., Tulmin H., Ehebauer M.T., Fernandez-Fuentes N., Russell A.J., Carr S.B., Phillips S.E., et. al.
Nature Communications,
2018
64.

Zhang Z., Gao R., Hu Q., Peacock H., Peacock D.M., Dai S., Shokat K.M., Suga H.
ACS Central Science,
2020
65.

Tanada M., Tamiya M., Matsuo A., Chiyoda A., Takano K., Ito T., Irie M., Kotake T., Takeyama R., Kawada H., Hayashi R., Ishikawa S., Nomura K., Furuichi N., Morita Y., et. al.
Journal of the American Chemical Society,
2023
66.

Nakajima E.C., Drezner N., Li X., Mishra-Kalyani P.S., Liu Y., Zhao H., Bi Y., Liu J., Rahman A., Wearne E., Ojofeitimi I., Hotaki L.T., Spillman D., Pazdur R., Beaver J.A., et. al.
Clinical Cancer Research,
2021
67.

Zhang Z., Shokat K.M.
Angewandte Chemie - International Edition,
2019
68.

Schulze C.J., Seamon K.J., Zhao Y., Yang Y.C., Cregg J., Kim D., Tomlinson A., Choy T.J., Wang Z., Sang B., Pourfarjam Y., Lucas J., Cuevas-Navarro A., Ayala-Santos C., Vides A., et. al.
Science,
2023
69.

Jiang J., Jiang L., Maldonato B.J., Wang Y., Holderfield M., Aronchik I., Winters I.P., Salman Z., Blaj C., Menard M., Brodbeck J., Chen Z., Wei X., Rosen M.J., Gindin Y., et. al.
Cancer Discovery,
2024
70.

Holderfield M., Lee B.J., Jiang J., Tomlinson A., Seamon K.J., Mira A., Patrucco E., Goodhart G., Dilly J., Gindin Y., Dinglasan N., Wang Y., Lai L.P., Cai S., Jiang L., et. al.
Nature,
2024
71.

Eliseev I.A., Shkil D.O., Shumakova V.S., Chernyshov V.V., Balakin K.V., Ivanov R.A.
Current Medicinal Chemistry,
2025